A Cure Exists. American Families Can’t Have It.

A Cure Exists. American Families Can’t Have It.
The Issue
AN OPEN LETTER TO THE COMMUNITY - Your support can save lives
Regarding Access to a Life-Saving Treatment for Children and AdultsLiving with Epstein-Barr Virus Positive Post-Transplant Lymphoproliferative Disease (EBV+ PTLD)
When your child receives a transplant, you celebrate. You breathe again. You start to imagine a future. And then, sometimes, the phone rings with news that no parent should ever have to hear: your child has cancer.
This is the reality for families living with EBV+ PTLD. It is a rare and deadly blood cancer that develops after transplantation, and it moves fast. For patients whose disease returns or fails to respond to initial treatment, there are no FDA-approved treatment options in the United States. Without effective treatment, survival is often measured in weeks to months. As parents, as families, as a community, we are writing to say: this is not acceptable.
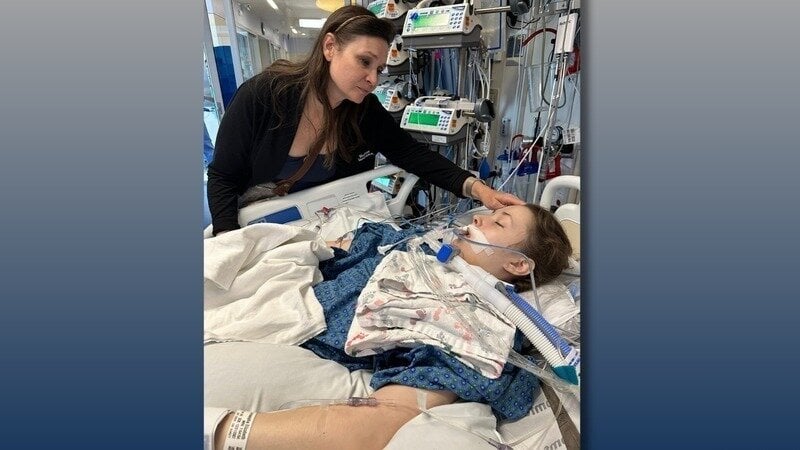
MEET ANNASOPHIA BERRY
AnnaSophia was born with complex congenital heart disease and received her first heart transplant as an infant. As a teenager, she developed chronic rejection and, in June 2025, received a second heart transplant after 48 days on the waiting list.
She survived. She began to recover. And then she was diagnosed with PTLD.
Her care team faced a choice that no family should have to witness: increase immunosuppression to protect the new heart, or decrease it to fight the cancer. Treating one condition could worsen the other.
AnnaSophia continues to be closely monitored for PTLD recurrence. Her family knows that if her disease progresses, timely access to an effective treatment will be critical. Right now, no such treatment is approved in the United States.
You can help AnnaSophia and the many families like hers right now. Sign this letter and add your voice to the call for a proven treatment to be made available in the United States. It takes only a moment, and it means everything to families who are out of time.
Read AnnaSophia's full story at https://www.transplantfamilies.org/post/fighting-for-her-heart-twice-annasophia-s-story
What Is EBV+ PTLD?
EBV+ PTLD is an ultra-rare, aggressive blood malignancy that can develop in people who have received a solid organ transplant or bone marrow transplant. The anti-rejection medications transplant recipients depend on to keep their new organ suppress the immune system, and in some patients, this allows the Epstein-Barr virus to go unchecked, leading to this devastating cancer.
It can affect anyone who has received a transplant, including very young children. For those whose disease returns after initial treatment, the prognosis without further intervention is dire. These are patients who already fought once to survive. They deserve the chance to fight again.
Without treatment, patients with relapsed or refractory EBV+ PTLD survive an average of only weeks to months.
With tabelecleucel, more than half of patients responded to treatment, and among those who responded, median survival had not yet been reached at nearly two years of follow-up. The difference between access and no access to this therapy is, quite literally, a matter of life and death.
A Treatment Exists. Just Not Here.
Here is something our community finds very hard to accept: a proven, effective treatment for relapsed or refractory EBV+ PTLD already exists. It is called tabelecleucel (EBVALLO™), and it has been approved and in use in Europe since December 2022, in the United Kingdom since May 2023, and in Switzerland since May 2024.
Families in those countries can access this therapy today. Families in the United States cannot.
Tabelecleucel is an innovative, off-the-shelf T-cell immunotherapy designed to target and eliminate EBV-infected cells. It does not require a matched donor. It is ready when patients need it. The clinical evidence behind it is substantial:
• More than 43 patients were treated in clinical studies, including children as young as two years old.
• More than half of patients responded to treatment, with over a quarter achieving a complete response.
• In children specifically, the response rate and safety profile were consistent with what was seen in adults.
• For patients who responded, many remained in remission for nearly two years or more.
• The safety profile was favorable, with no treatment-related fatalities, no graft rejection, and no cytokine release syndrome.
Why Existing Options Are Not Enough
Parents often ask: aren't there other treatments available? The honest answer is that there are options, but none were designed for this disease, none are FDA-approved for it, and all carry serious trade-offs that make them especially difficult for transplant patients.
Reducing immunosuppression is almost always the first step. By dialing back anti-rejection medication, the immune system may recover enough to fight the cancer. But doing so puts the transplanted organ at risk of rejection. Families are forced into an impossible choice between the cancer and the organ their child depends on to survive.
Rituximab and chemotherapy are the standard next steps, but many patients relapse or do not respond. For those patients, there is nothing FDA-approved left to offer. Chemotherapy is also particularly difficult in this population because transplant recipients are already vulnerable to infection and organ toxicity.
Donor-derived T-cell infusions can work for some bone marrow transplant patients, but they require a suitable donor match and are only available at a small number of specialized research centers. They are also far more limited for solid organ transplant patients, which includes most children with transplant-related PTLD.
Tabelecleucel was designed to fill precisely this gap. It does not require a matched donor, is available off-the-shelf when a patient needs it, was built specifically for EBV+ PTLD, and has a safety profile that includes no cases of graft rejection in clinical trials. For a population whose organ survival depends on careful immune management, that last point is not a footnote. It is everything.
What Happened at the FDA
The path to FDA approval has been painful and confusing to watch as families who needed this therapy. The application for tabelecleucel was submitted to the FDA with data from 43 patients. The FDA granted it Priority Review status and Accelerated Approval status, recognizing the urgent unmet need. Researchers and the FDA engaged in detailed dialogue about the clinical trial design over more than five years.
In January 2025, the FDA issued its first rejection letter, citing a manufacturing facility inspection issue. No safety or efficacy problems were found. The developers resolved the issue and resubmitted. Priority Review was granted again, with a January 10, 2026 decision date.
On January 9, 2026, a second rejection letter arrived. This time, the FDA reversed its long-standing position, stating that the clinical trial design it had previously accepted over years of back-and-forth was no longer adequate. No new safety problems were raised. No new clinical deficiencies were identified. The manufacturing issue had been resolved. The FDA simply changed course.
What concerns our community is this: the reversal appears to reflect a change in the review team at the FDA rather than a change in the evidence. When the people reviewing an application change, families with rare diseases cannot afford for the goalposts to move with them. Our children are not statistical edge cases. They are real, and they are running out of time. We are not asking the FDA to lower its standards. We are asking it to honor the process it established and ensure that patients have a clear path to access this therapy while any remaining questions are resolved.
What We Are Asking
As a community of patients, families, and advocates, we are calling on the FDA to:
• Urgently engage with the developers of tabelecleucel to establish a clear, achievable, and expedited path to patient access, whether through approval or accelerated approval.
• Recognize and respect the regulatory decisions made by the European Commission, the UK MHRA, and Swissmedic, all of which found the clinical evidence sufficient to approve this therapy.
• Apply evidence standards that are appropriate for ultra-rare pediatric diseases, where large randomized trials are often not feasible and where time is a factor that cannot be ignored.
• Ensure that families who need access now are supported through a robust and accessible Expanded Access Program while the approval process continues.
What You Can Do
Rare diseases are often invisible to the public because so few people have heard of them. You can change that. The families facing EBV+ PTLD are not asking for special treatment. They are asking for the same access to proven medicine that families in Europe already have.
• Sign this letter and share it. Every signature is a voice telling the FDA that this community is watching and that these patients matter.
• Share this letter and the stories of PTLD families on social media. Use #EBVPTLDCantWait to help build awareness.
• If you work in medicine, research, or patient advocacy, add your name and your professional voice.
• Help your community understand what EBV+ PTLD is and why access to treatment matters. Education is the foundation of change.
These children have already been through so much. A transplant is supposed to be a second chance at life. EBV+ PTLD threatens to take that chance away. A proven treatment exists. It is available to patients in other countries right now. We are asking that U.S. families have the same opportunity. Sign this letter. Share it with everyone you know. Help us make sure no family faces this disease without options.
Signed,
Melissa McQueen, MBA - Executive Director, Transplant Families
Brittany Clayborne, PsyD - Executive Director, Transplant Teenz
Organizations
Rick Lofgren, MBA - President, Children’s Organ Transplant Association (COTA)
Jen Benson - The Transplant Journey
Jordan Sarbaugh - President, BARE
Roberta Smith - President, Alagille Syndrome Alliance (ALGSA)
Vicky Ng, MD - President, North American Society for Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN)
Mandy Sandkhuler - Director of Partnerships and Communications, The Mended Hearts, Inc.
Leah Mumm, M.S. - Director of Family and Physician Relations, Children’s Cardiomyopathy Foundation
Jenny Hsieh - President, Danon Disease Foundation
Lorraine Tallman - CEO and Founder, Amanda Hope Rainbow Angels
Steven Coffee, MA, EMCQSL - CEO, Head2HeartConnections LLC
Edward Drake II - Founder, The YNOTT? Foundation
Individual Signatories
Simon Horslen MB ChB FRCPCH FAST FAASLD
Pediatric Transplant Hepatologist
Taylor Heald-Sargent, MD PhD
Pediatric Transplant Infectious Disease
Amber Hildreth, DO, FAAP
Pediatric Transplant Hepatologist

118
The Issue
AN OPEN LETTER TO THE COMMUNITY - Your support can save lives
Regarding Access to a Life-Saving Treatment for Children and AdultsLiving with Epstein-Barr Virus Positive Post-Transplant Lymphoproliferative Disease (EBV+ PTLD)
When your child receives a transplant, you celebrate. You breathe again. You start to imagine a future. And then, sometimes, the phone rings with news that no parent should ever have to hear: your child has cancer.
This is the reality for families living with EBV+ PTLD. It is a rare and deadly blood cancer that develops after transplantation, and it moves fast. For patients whose disease returns or fails to respond to initial treatment, there are no FDA-approved treatment options in the United States. Without effective treatment, survival is often measured in weeks to months. As parents, as families, as a community, we are writing to say: this is not acceptable.
MEET ANNASOPHIA BERRY
AnnaSophia was born with complex congenital heart disease and received her first heart transplant as an infant. As a teenager, she developed chronic rejection and, in June 2025, received a second heart transplant after 48 days on the waiting list.
She survived. She began to recover. And then she was diagnosed with PTLD.
Her care team faced a choice that no family should have to witness: increase immunosuppression to protect the new heart, or decrease it to fight the cancer. Treating one condition could worsen the other.
AnnaSophia continues to be closely monitored for PTLD recurrence. Her family knows that if her disease progresses, timely access to an effective treatment will be critical. Right now, no such treatment is approved in the United States.
You can help AnnaSophia and the many families like hers right now. Sign this letter and add your voice to the call for a proven treatment to be made available in the United States. It takes only a moment, and it means everything to families who are out of time.
Read AnnaSophia's full story at https://www.transplantfamilies.org/post/fighting-for-her-heart-twice-annasophia-s-story
What Is EBV+ PTLD?
EBV+ PTLD is an ultra-rare, aggressive blood malignancy that can develop in people who have received a solid organ transplant or bone marrow transplant. The anti-rejection medications transplant recipients depend on to keep their new organ suppress the immune system, and in some patients, this allows the Epstein-Barr virus to go unchecked, leading to this devastating cancer.
It can affect anyone who has received a transplant, including very young children. For those whose disease returns after initial treatment, the prognosis without further intervention is dire. These are patients who already fought once to survive. They deserve the chance to fight again.
Without treatment, patients with relapsed or refractory EBV+ PTLD survive an average of only weeks to months.
With tabelecleucel, more than half of patients responded to treatment, and among those who responded, median survival had not yet been reached at nearly two years of follow-up. The difference between access and no access to this therapy is, quite literally, a matter of life and death.
A Treatment Exists. Just Not Here.
Here is something our community finds very hard to accept: a proven, effective treatment for relapsed or refractory EBV+ PTLD already exists. It is called tabelecleucel (EBVALLO™), and it has been approved and in use in Europe since December 2022, in the United Kingdom since May 2023, and in Switzerland since May 2024.
Families in those countries can access this therapy today. Families in the United States cannot.
Tabelecleucel is an innovative, off-the-shelf T-cell immunotherapy designed to target and eliminate EBV-infected cells. It does not require a matched donor. It is ready when patients need it. The clinical evidence behind it is substantial:
• More than 43 patients were treated in clinical studies, including children as young as two years old.
• More than half of patients responded to treatment, with over a quarter achieving a complete response.
• In children specifically, the response rate and safety profile were consistent with what was seen in adults.
• For patients who responded, many remained in remission for nearly two years or more.
• The safety profile was favorable, with no treatment-related fatalities, no graft rejection, and no cytokine release syndrome.
Why Existing Options Are Not Enough
Parents often ask: aren't there other treatments available? The honest answer is that there are options, but none were designed for this disease, none are FDA-approved for it, and all carry serious trade-offs that make them especially difficult for transplant patients.
Reducing immunosuppression is almost always the first step. By dialing back anti-rejection medication, the immune system may recover enough to fight the cancer. But doing so puts the transplanted organ at risk of rejection. Families are forced into an impossible choice between the cancer and the organ their child depends on to survive.
Rituximab and chemotherapy are the standard next steps, but many patients relapse or do not respond. For those patients, there is nothing FDA-approved left to offer. Chemotherapy is also particularly difficult in this population because transplant recipients are already vulnerable to infection and organ toxicity.
Donor-derived T-cell infusions can work for some bone marrow transplant patients, but they require a suitable donor match and are only available at a small number of specialized research centers. They are also far more limited for solid organ transplant patients, which includes most children with transplant-related PTLD.
Tabelecleucel was designed to fill precisely this gap. It does not require a matched donor, is available off-the-shelf when a patient needs it, was built specifically for EBV+ PTLD, and has a safety profile that includes no cases of graft rejection in clinical trials. For a population whose organ survival depends on careful immune management, that last point is not a footnote. It is everything.
What Happened at the FDA
The path to FDA approval has been painful and confusing to watch as families who needed this therapy. The application for tabelecleucel was submitted to the FDA with data from 43 patients. The FDA granted it Priority Review status and Accelerated Approval status, recognizing the urgent unmet need. Researchers and the FDA engaged in detailed dialogue about the clinical trial design over more than five years.
In January 2025, the FDA issued its first rejection letter, citing a manufacturing facility inspection issue. No safety or efficacy problems were found. The developers resolved the issue and resubmitted. Priority Review was granted again, with a January 10, 2026 decision date.
On January 9, 2026, a second rejection letter arrived. This time, the FDA reversed its long-standing position, stating that the clinical trial design it had previously accepted over years of back-and-forth was no longer adequate. No new safety problems were raised. No new clinical deficiencies were identified. The manufacturing issue had been resolved. The FDA simply changed course.
What concerns our community is this: the reversal appears to reflect a change in the review team at the FDA rather than a change in the evidence. When the people reviewing an application change, families with rare diseases cannot afford for the goalposts to move with them. Our children are not statistical edge cases. They are real, and they are running out of time. We are not asking the FDA to lower its standards. We are asking it to honor the process it established and ensure that patients have a clear path to access this therapy while any remaining questions are resolved.
What We Are Asking
As a community of patients, families, and advocates, we are calling on the FDA to:
• Urgently engage with the developers of tabelecleucel to establish a clear, achievable, and expedited path to patient access, whether through approval or accelerated approval.
• Recognize and respect the regulatory decisions made by the European Commission, the UK MHRA, and Swissmedic, all of which found the clinical evidence sufficient to approve this therapy.
• Apply evidence standards that are appropriate for ultra-rare pediatric diseases, where large randomized trials are often not feasible and where time is a factor that cannot be ignored.
• Ensure that families who need access now are supported through a robust and accessible Expanded Access Program while the approval process continues.
What You Can Do
Rare diseases are often invisible to the public because so few people have heard of them. You can change that. The families facing EBV+ PTLD are not asking for special treatment. They are asking for the same access to proven medicine that families in Europe already have.
• Sign this letter and share it. Every signature is a voice telling the FDA that this community is watching and that these patients matter.
• Share this letter and the stories of PTLD families on social media. Use #EBVPTLDCantWait to help build awareness.
• If you work in medicine, research, or patient advocacy, add your name and your professional voice.
• Help your community understand what EBV+ PTLD is and why access to treatment matters. Education is the foundation of change.
These children have already been through so much. A transplant is supposed to be a second chance at life. EBV+ PTLD threatens to take that chance away. A proven treatment exists. It is available to patients in other countries right now. We are asking that U.S. families have the same opportunity. Sign this letter. Share it with everyone you know. Help us make sure no family faces this disease without options.
Signed,
Melissa McQueen, MBA - Executive Director, Transplant Families
Brittany Clayborne, PsyD - Executive Director, Transplant Teenz
Organizations
Rick Lofgren, MBA - President, Children’s Organ Transplant Association (COTA)
Jen Benson - The Transplant Journey
Jordan Sarbaugh - President, BARE
Roberta Smith - President, Alagille Syndrome Alliance (ALGSA)
Vicky Ng, MD - President, North American Society for Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN)
Mandy Sandkhuler - Director of Partnerships and Communications, The Mended Hearts, Inc.
Leah Mumm, M.S. - Director of Family and Physician Relations, Children’s Cardiomyopathy Foundation
Jenny Hsieh - President, Danon Disease Foundation
Lorraine Tallman - CEO and Founder, Amanda Hope Rainbow Angels
Steven Coffee, MA, EMCQSL - CEO, Head2HeartConnections LLC
Edward Drake II - Founder, The YNOTT? Foundation
Individual Signatories
Simon Horslen MB ChB FRCPCH FAST FAASLD
Pediatric Transplant Hepatologist
Taylor Heald-Sargent, MD PhD
Pediatric Transplant Infectious Disease
Amber Hildreth, DO, FAAP
Pediatric Transplant Hepatologist

118
Petition Updates
Share this petition
Petition created on April 8, 2026