

Ensure equitable access to molecular testing for biliary tract cancers in Ontario


Ensure equitable access to molecular testing for biliary tract cancers in Ontario
The Issue
Summary:
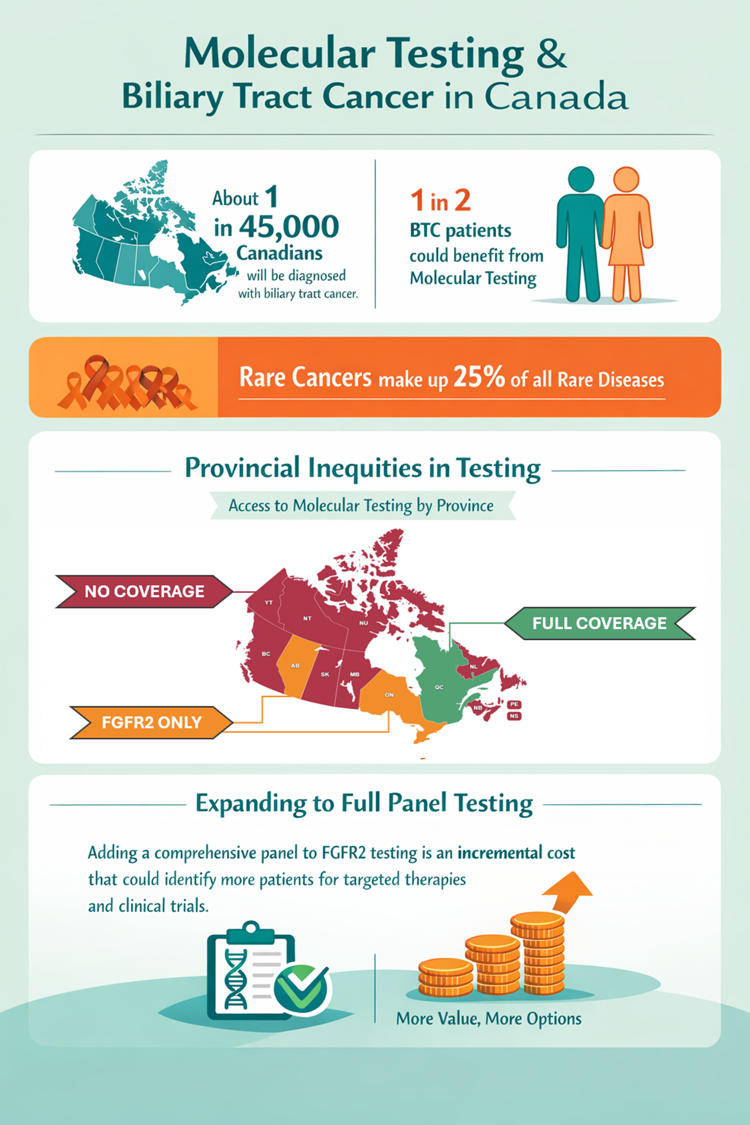
As of January 1, 2026, most Canadians with biliary tract cancer have lost funded access to essential molecular testing, despite strong clinical guideline recommendations and evidence that up to 50 percent of patients could benefit.
Without testing, patients with advanced disease that has spread are often limited to standard chemotherapy, where median survival is typically ~12 months. Molecular testing provides critical information about a tumour’s biology, enabling more personalized treatment options and access to targeted therapies and clinical trials which can extend survival and significantly improve quality of life.
This petition calls on provinces to recognize comprehensive molecular testing as essential care and cover the cost of testing for late stage biliary tract cancer patients.
What is Molecular Testing?
Molecular testing, also known as Biomarker testing or Genomic sequencing, can open the door to personalized treatment options and clinical trials. It involves examining genetic material usually from a tumour biopsy, to identify mutations and alterations known to be associated with cholangiocarcinoma. Identifying which biomarkers are present can enable physicians to tailor their treatment to the individual’s tumour biology, avoiding ineffective therapies and providing additional targeted treatment options once the standard of care has been exhausted. Some common tumour markers are FGFR2 fusions, IDH1 and BRCA1/2 mutations – all of which have targeted agents.
Why does Molecular Testing matter?
This issue affects everyone involved in cancer care. For patients who cannot access molecular testing, treatment is often limited to standard chemotherapy, where median survival at diagnosis can be as short as six months. Molecular testing can identify targeted therapies and clinical trials that may extend survival by years for many patients, while also significantly improving quality of life.
All major clinical guidelines recommend molecular testing for biliary tract cancer, yet many Canadians cannot access it. This is equivalent to giving clinicians a roadmap for optimal care while withholding the tools needed to follow it.
Most clinical trials require molecular testing prior to enrolment. Routine, publicly funded testing would improve trial recruitment and help attract companies with molecularly targeted therapies to open trials in Canada.
Molecular testing also generates critical real-world evidence, which is severely lacking for this rare cancer population. Comprehensive data strengthens research, informs future therapies, and supports Canada’s ability to deliver modern precision oncology care.
Barriers in Canada that limit access to molecular testing:
- Financial barriers, where many patients must personally pay up to $5000
- Inadequate or incomplete testing, resulting in wasted time and loss of valuable tissue samples.
- Geographic inequities, where testing is funded in some regions but unavailable in others.
- Administrative uncertainty, where access depends on fragmented programs vs policy-backed funding.
Our Strategy
This petition calls on policymakers to recognize molecular testing as essential care for people with biliary tract cancers. Because molecular testing is funded at the provincial level in Canada, our strategy is to advocate province by province, starting with Ontario.
Ontario represents approximately 40 percent of Canada’s population and already funds molecular testing for a single biomarker (FGFR2). Expanding this to include broader gene panel testing would require only an incremental increase in cost, while significantly improving the ability to guide treatment decisions and identify eligibility for clinical trials.
Our goal is to see comprehensive molecular testing become part of the standard of care for biliary tract cancer patients. Testing should be performed as early as possible after diagnosis so patients and care teams can understand what treatment options may be available and avoid unnecessary delays in care.
Why Sign the Petition?
Whether you are a patient, caregiver, clinician, or ally, your signature matters. Signing this petition will help ensure molecular testing is treated as essential care, not a privilege, for Canadians with biliary tract cancers.
-----------------------------------------------------------------------------
1,352
The Issue
Summary:
As of January 1, 2026, most Canadians with biliary tract cancer have lost funded access to essential molecular testing, despite strong clinical guideline recommendations and evidence that up to 50 percent of patients could benefit.
Without testing, patients with advanced disease that has spread are often limited to standard chemotherapy, where median survival is typically ~12 months. Molecular testing provides critical information about a tumour’s biology, enabling more personalized treatment options and access to targeted therapies and clinical trials which can extend survival and significantly improve quality of life.
This petition calls on provinces to recognize comprehensive molecular testing as essential care and cover the cost of testing for late stage biliary tract cancer patients.
What is Molecular Testing?
Molecular testing, also known as Biomarker testing or Genomic sequencing, can open the door to personalized treatment options and clinical trials. It involves examining genetic material usually from a tumour biopsy, to identify mutations and alterations known to be associated with cholangiocarcinoma. Identifying which biomarkers are present can enable physicians to tailor their treatment to the individual’s tumour biology, avoiding ineffective therapies and providing additional targeted treatment options once the standard of care has been exhausted. Some common tumour markers are FGFR2 fusions, IDH1 and BRCA1/2 mutations – all of which have targeted agents.
Why does Molecular Testing matter?
This issue affects everyone involved in cancer care. For patients who cannot access molecular testing, treatment is often limited to standard chemotherapy, where median survival at diagnosis can be as short as six months. Molecular testing can identify targeted therapies and clinical trials that may extend survival by years for many patients, while also significantly improving quality of life.
All major clinical guidelines recommend molecular testing for biliary tract cancer, yet many Canadians cannot access it. This is equivalent to giving clinicians a roadmap for optimal care while withholding the tools needed to follow it.
Most clinical trials require molecular testing prior to enrolment. Routine, publicly funded testing would improve trial recruitment and help attract companies with molecularly targeted therapies to open trials in Canada.
Molecular testing also generates critical real-world evidence, which is severely lacking for this rare cancer population. Comprehensive data strengthens research, informs future therapies, and supports Canada’s ability to deliver modern precision oncology care.
Barriers in Canada that limit access to molecular testing:
- Financial barriers, where many patients must personally pay up to $5000
- Inadequate or incomplete testing, resulting in wasted time and loss of valuable tissue samples.
- Geographic inequities, where testing is funded in some regions but unavailable in others.
- Administrative uncertainty, where access depends on fragmented programs vs policy-backed funding.
Our Strategy
This petition calls on policymakers to recognize molecular testing as essential care for people with biliary tract cancers. Because molecular testing is funded at the provincial level in Canada, our strategy is to advocate province by province, starting with Ontario.
Ontario represents approximately 40 percent of Canada’s population and already funds molecular testing for a single biomarker (FGFR2). Expanding this to include broader gene panel testing would require only an incremental increase in cost, while significantly improving the ability to guide treatment decisions and identify eligibility for clinical trials.
Our goal is to see comprehensive molecular testing become part of the standard of care for biliary tract cancer patients. Testing should be performed as early as possible after diagnosis so patients and care teams can understand what treatment options may be available and avoid unnecessary delays in care.
Why Sign the Petition?
Whether you are a patient, caregiver, clinician, or ally, your signature matters. Signing this petition will help ensure molecular testing is treated as essential care, not a privilege, for Canadians with biliary tract cancers.
-----------------------------------------------------------------------------

1,352
Supporter Voices
Petition Updates
Share this petition
Petition created on February 10, 2026