

#Fight4Hemovigilance: Save Canada's Blood Safety Contribution Program


#Fight4Hemovigilance: Save Canada's Blood Safety Contribution Program
The Issue
Dear Decision-Makers,
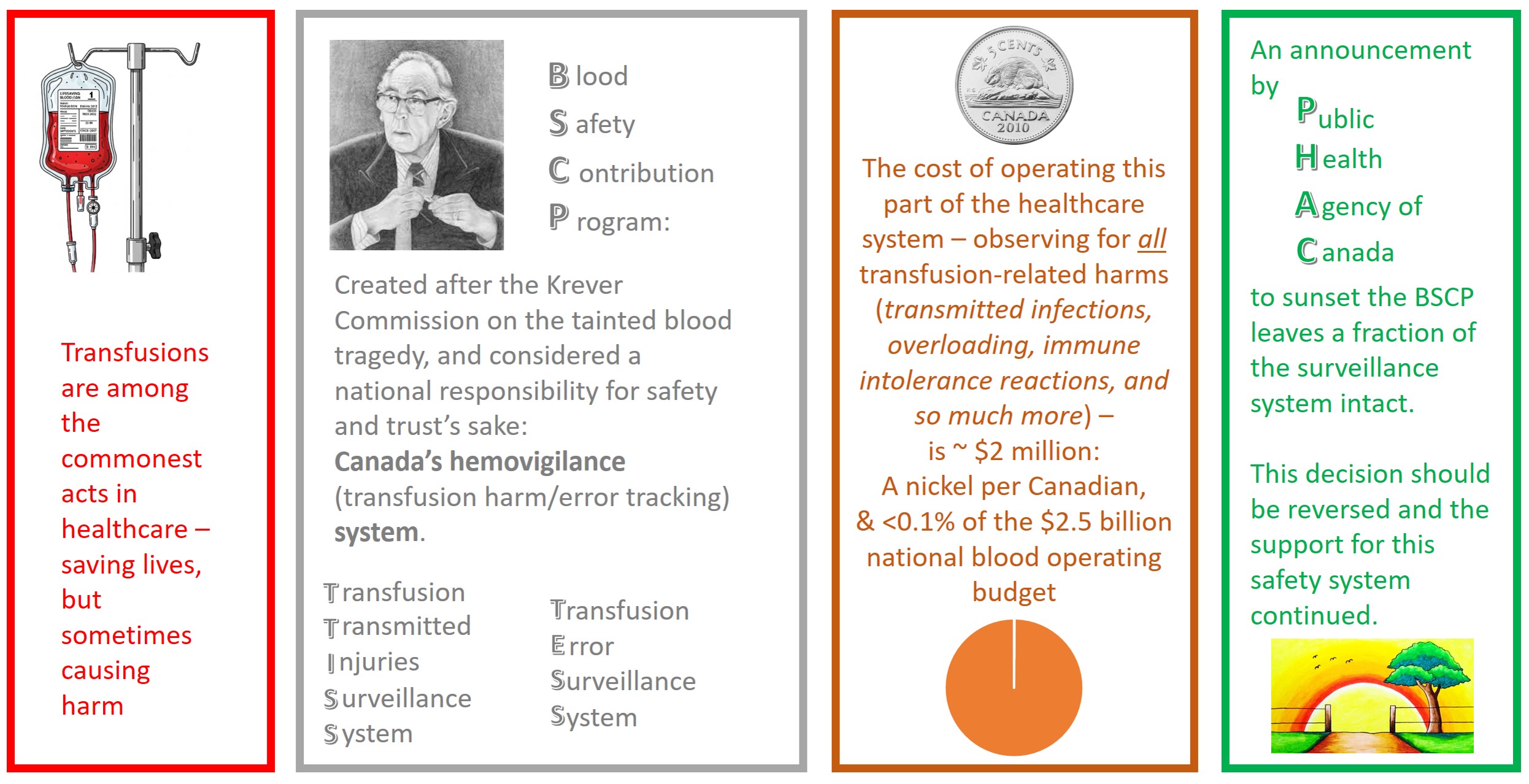
I ask that the Public Health Agency of Canada (PHAC) rescind its decision to sunset the Blood Safety Contribution Program (BSCP) – namely its Transfusion Transmitted Injuries Surveillance System (TTISS) and the Transfusion Errors Surveillance System (TESS) – effective April 1st, 2026 (as the arbitrarily-selected de-activation date).
This unilateral decision was issued on August 28th, 2024 without engagement of patients or the nation-wide transfusion service community supporting them. Only a handful of those who directly contribute data to the programs were notified. Lacking any avenues for meaningful negotiation or permissions for fan-out, this was an effective silencing of potential dissent and action.
On its website in the months that followed, PHAC did not reveal its decision, though the (anonymized) letter was ultimately exposed by the counter-action of the Canadian Society for Transfusion Medicine (CSTM) in their Hemovigilance Under Threat webpage https://transfusion.ca/our-work/hemovigilance/ ; letter: https://transfusion.ca/wp-content/uploads/2025/12/PHAC-Sunsetting-Announcement-De-identified.pdf on May 28th, 2025 prior to their annual conference.
A Call-to-Action open letter by Shih & Prokopchuk-Gauk, https://nacblood.ca/en/resource/nac-letter-cbs-ptblc-urgent-need-transform-canadian-hemovigilance-system-call-action from the National Advisory Committee (NAC) articulated numerous concerns on this decision.
On July 25, 2025, the leaders of Canada’s blood systems—Dr. Graham Sher of Canadian Blood Services and Nathalie Fagnan of Héma‑Québec—issued a joint letter urging reconsideration of the decision to close the BSCP. They proposed measures to address the findings that led to the shutdown plan, and called for a moratorium on decommissioning steps until formal discussions and actions on void management are successfully completed. https://www.transfusion.ca/getmedia/bc7e5d70-55c1-4163-8875-afa69cfa2349/2025-07-25_Letter_CEOs-to-PHAC-President-Nancy-Hamzawi_re_Consensus-Conference_EN.pdf.aspx
If the data infrastructure, its holdings, and the staffing of TTISS are altogether made obsolete, we are left to rely on the residual Blood Regulations in the Canada Vigilance Program (CVP) for counting a more limited range of adverse events (AE) or errors/accidents (E/A) of interest. The focus drops to container-side (supplier-relevant) concerns rather than to the full end-to-end process of transfusion safety and/or to the capture of extraordinary breakthrough injuries unique to a product-patient interaction (however unimpeachable the donor/blood and transfusion safety practices might have been).
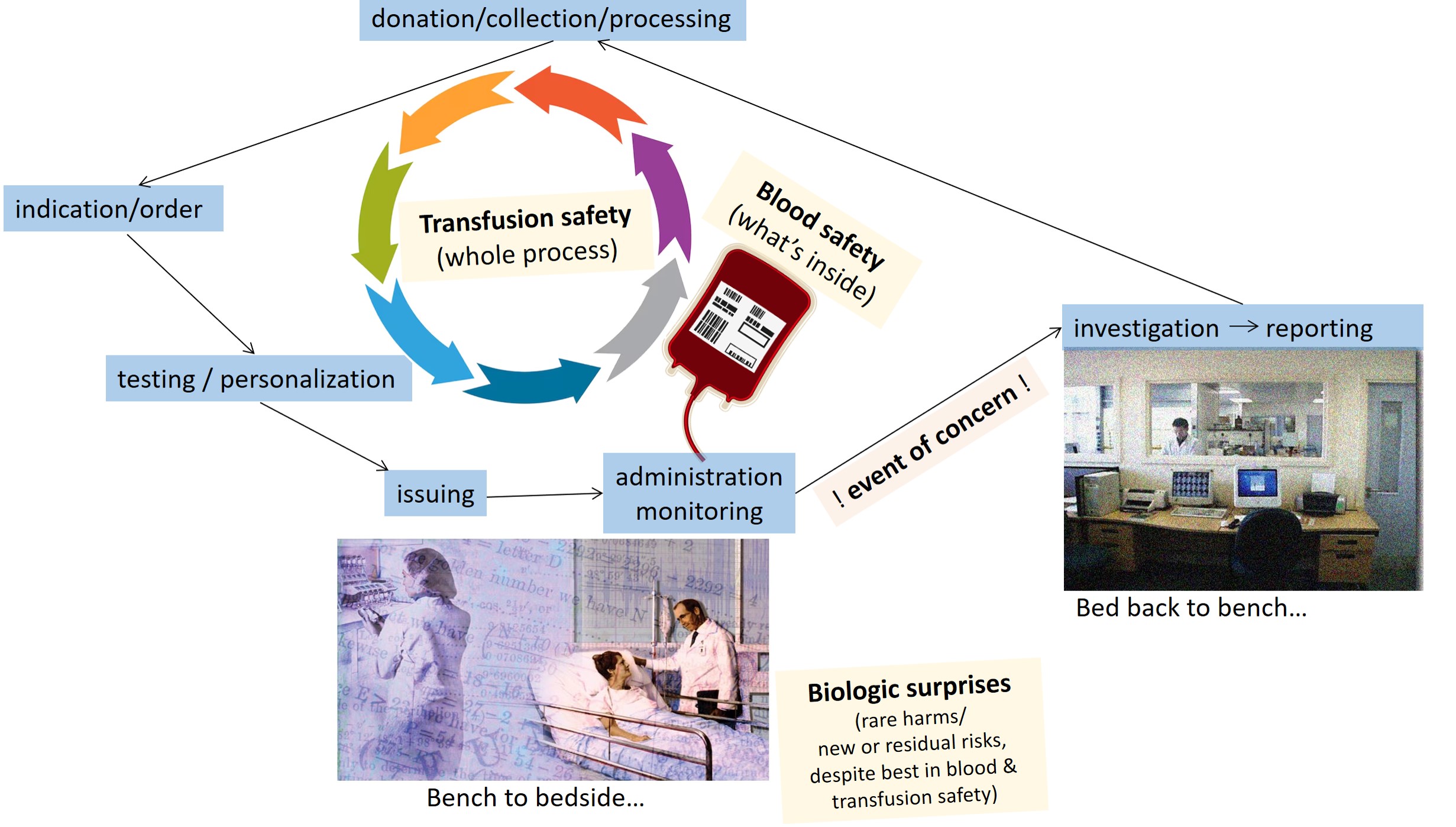
Put simply, we are violating our Public Health responsibility to sense known or emerging harms by this top-down devaluation.
This betrays public trust, while backsliding on the global stage of meeting basic transfusion safety standards.
Hemovigilance aims to capture the observed harms of transfusion to make this common form of care safer. We code for what the harm was, its severity, the persistence of an injury’s effects (for those who survive), and for whether the event has implications for harm to others (ie- were there missed clues in the past; are there current containers to quarantine or recall now; and are there changes to future donor management or production practices).
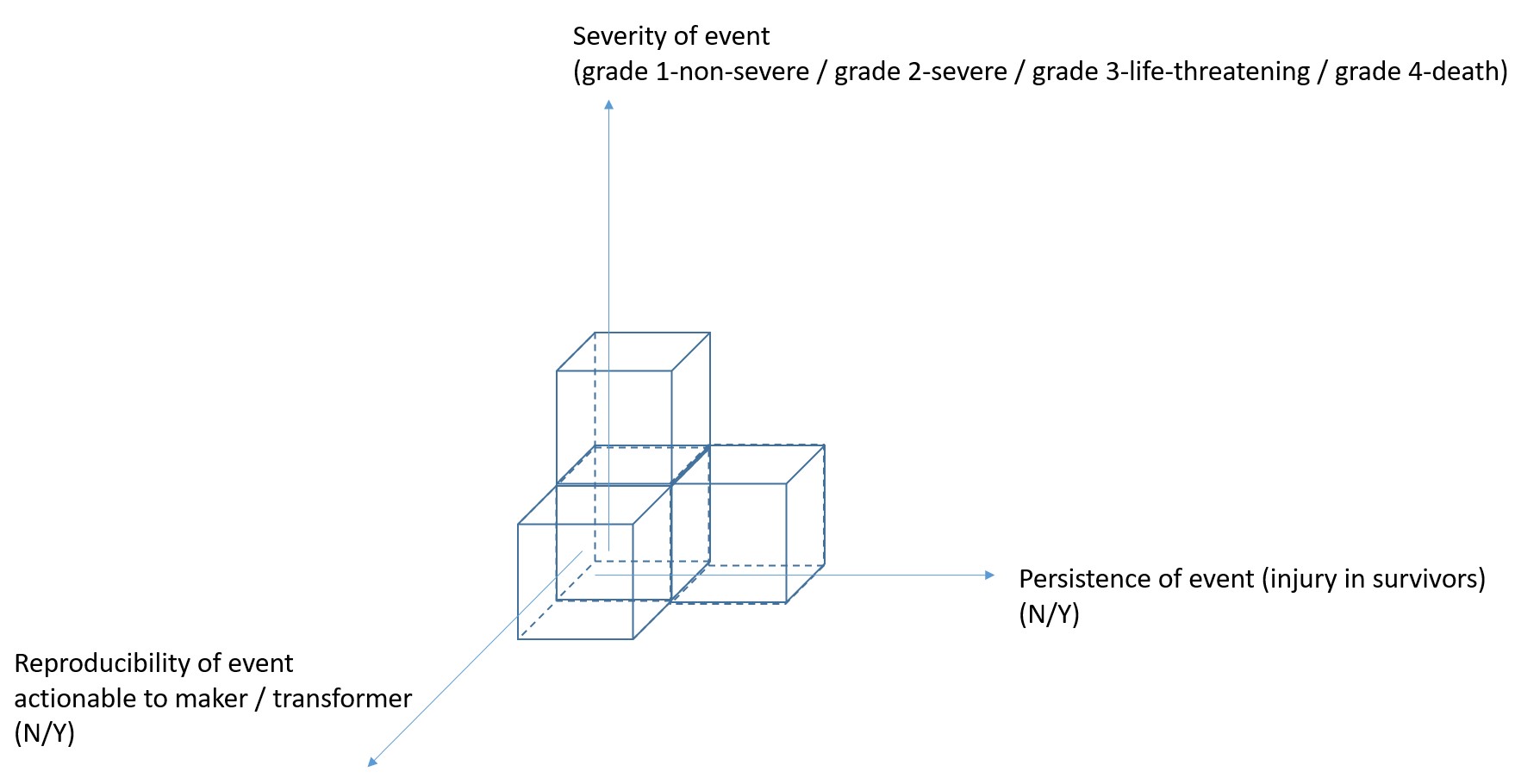
The blood operators/manufacturers and the CVP have coordinated well on many “3D hits” in blood quality, be it a transfusion transmitted infection (TTI) or another reliably toxic factor.
However – not one of these core bodies is looking comprehensively at all the other now dominant and enduring harms that TTISS has been tracking. While not usually related to specific donors or modes of production/preparation, these are some of the greatest threats appreciated in transfusion care today (be it by frequency, severity, and/or by the power to prey upon the most fragile of our patients).
#1: Transfusion-associated circulatory overload (TACO). [1] Globally, this is the #1 cause of transfusion-related death. When TACO is re-scaled for under-reporting, it is also among the commonest of transfusion reactions (at a 1-10% rate per blood encounter). TACO is notoriously “disposition-escalating” (ie- summoning critical care teams or transfers [in 20% of cases][2]), and protracted in its effects (ie- extending hospital lengths of stay).
TACO is of such importance in the eyes of laboratory assessors that it has been the subject of prime inspection (per Bulletins on the expectation that policies identify those at risk and provide mitigation options).[3] TACO reflects problems upstream of the integrity of what sits within the container, and there are greater clinician, systems, and patient pain-points driving this often preventable event. TACO targets those with already poor heart-lung function, with the most susceptible at the age-extremes (the smallest, sickest, and/or oldest of our patients).[4] Closing TTISS and TESS is a curtain call for knowing the counts, understanding contributors (eg- under-resourced wards [insufficiently-examined patients, poorly paced infusions]), and designing course-corrections.
#2: HyperHemolysis Syndrome (HHS) as a particular kind of Delayed Hemolytic Transfusion Reaction (DHTR).
Tracking with the devastation of sickle cell disease (SCD) are the compounding weights of systemic racism and complex blood biology (ie- red and white cell co-adaptive variants selected by tropical infectious diseases like malaria). The latter variants can make transfusions trickier, no matter how meticulous our matching approach may be.
HyperHemolysis Syndrome (HHS), an unexpected immune over-reaction to transfusion, leaves a blood recipient more anemic than where they started. Red cell destruction goes beyond the offending unit with demolition of the patient’s own red cells, while the power to compensate is also undermined. A multi-system injury, despite intensive care, may nevertheless end in death. At hospitals taking care of people with SCD, fatalities from HHS can be on par with deaths from TACO or non-overload transfusion-related acute lung injury (TRALI).
If TTISS closes, there is no one left to keep central count of HHS.
This contravenes all united calls by exemplar hemovigilance systems, such as the Serious Hazards of Transfusion in the United Kingdom [SHOT-UK] www.shotuk.org/wp-content/uploads/2024/10/SHOT-Bite-No.15-Hyperhaemolysis.pdf and advocates within the AABB. https://www.aabb.org/news-resources/news/article/2023/10/16/aabb23--hyperhemolysis-syndrome-presents-challenges
Without learning as much as we can from each case and their collective features, we cannot make organized progress in this already-marginalized population.
To suppress hemovigilance of HHS is to amplify rather than redress a major existing inequity.
The reporting of red cell rejection events across a spectrum – from minor delayed serologic transfusion reactions (DSTRs) (asymptomatic antibody responses), to delayed hemolytic transfusion reactions (DHTRs) (partial to complete anti-unit effects), to HHS (life-threatening bystander cellular effects) – has illuminated the impacts of insufficient laboratory information centralization as well. TTISS has been a repository for this continuum of reactions, and has therefore been a systems-integration "indicator."
The failure of laboratory information handover allows the unintended gamble of provocative re-exposure to an offending antigen in a next transfusion. The strategic neglect of healthcare fragmentation-related transfusion injuries is a means by which to suppress evidence for investing in better health information technology and its inter-operability.
#3: Other events not related to product quality, but rather to the biology of the host, and/or host-donor (idiosyncratic) interactions:
Severe, high case-fatality-rate adverse events such as post-transfusion purpura (PTP) and transfusion-associated graft versus host disease (TA-GVHD), when they are not due to operator or dispensation service failures, also go silent if TTISS closes.
How do we count the emerging concern of possible cerebral amyloid angiopathy?[5]
A national loss in tabulation-power for any such reported events-of-concern is incomprehensible to jurisdictions with developed hemovigilance systems.
The operating costs of the BSCP have not only been trivial in the context of overall government spending (ie- at just over $2million CAD annually – a mere nickel for each Canadian), but diminishing over time. Participating hospitals have long financed the largest part of their own reporting workload.
Relative to the costs of the entire blood system (at >$2billion CAD annually), TTISS costs < 0.1% – a figure that is also half of what it once was.
TTISS, despite its progressive underfunding and now the existential threat against it, gets credit for being the most informative data house (relative to the federal offices that would remain).
TTISS, operated (as with the health care systems) on the provincial and territorial level, has its very origins rooted in the evidence and recommendations of Horace Justice Krever in the Royal Commission of Inquiry on the Blood System in Canada (“the Krever Commission”).
As the nation sits upon literal mountains of data, and just as visions of better-connected, vein-to-vein Big Data are dawning with the promise of new analytics and enhanced insights in personalization,[6] the system is nearing elimination rather than enhancement.
Many Canadians still recall the tainted global blood tragedy.
Dr Graham Sher, CEO of Canadian Blood Services, wrote this at the passing of Krever 2 years ago in 2023:

Granted the triumphs against viral TTIs, and improved platelet and plasma safety by the contemporary modifications of pathogen reduction and additive solutions, would Krever agree that we have made such strides that a secretive shut-down of the BSCP by PHAC is justified?
How or why are other less-evidence-powered, non-commission-inspired programs permitted greater inefficiencies or investments?
How do we square the moves of an allegedly pro-healthcare administration with the playbook tactics of more conservative/opposition-party platforms that the majority of Canadians voted against?
I agree with the NAC Letter in the call to conserve (or enhance) hemovigilance funding, with a commitment to improve our system by reorganization, rather than by scripting its collapse.
This would align better with (rather than thwart) the recommendations on sharpening roles and responsibilities, with more timely analytic turnarounds, per the evaluation report of 2023. https://www.canada.ca/en/public-health/corporate/transparency/corporate-management-reporting/evaluation/blood-safety-contribution-program-2017-2018-2021-2022.html
Dismantling event or error/incident reporting systems is deeply counterproductive at a time when jurisdictions are more attuned than ever to these risks and the imperative to mitigate them.
As a fellow patriotic Canadian, each of us with a lifetime odds of 1-in-2 for needing a blood transfusion, I trust that we want what is safest for each other and our loved ones.
Let us save hemovigilance in Canada.
References:
1. Bulle EB, Klanderman RB, Pendergrast J, Cserti-Gazdewich C, Callum J, Vlaar APJ. The recipe for TACO: A narrative review on the pathophysiology and potential mitigation strategies of transfusion-associated circulatory overload. Blood Rev. 2022;52:100891. Epub 20211002. doi: 10.1016/j.blre.2021.100891. PubMed PMID: 34627651.
2. Lieberman L, Maskens C, Cserti-Gazdewich C, Hansen M, Lin Y, Pendergrast J, et al. A retrospective review of patient factors, transfusion practices, and outcomes in patients with transfusion-associated circulatory overload. Transfus Med Rev. 2013;27(4):206-12. Epub 20130926. doi: 10.1016/j.tmrv.2013.07.002. PubMed PMID: 24075097.
3. American Association of Blood Banks - Association Bulletin #15-02: Transfusion Associated Circulatory Overload (TACO). 2015 [updated 12 February 2016]. 15]. Available from: http://www.aabb.org/programs/publications/bulletins/Docs/ab15-02.pdf#search=bulletin%20%2315%2D02
4. McVey MJ, Cohen R, Arsenault V, Escorcia A, Tasmin F, Pendergrast J, et al. Frequency and timing of all-cause deaths in visits involving suspected transfusion reactions, and the significance of cardiopulmonary disturbances. Vox Sang. 2021;116(8):898-909. Epub 20210226. doi: 10.1111/vox.13086. PubMed PMID: 33634884.
5. Muir RT, Callum JL, Yu AYX, Kapral MK, Swartz RH, Black SE, et al. Beta-Amyloid Related Neurodegenerative and Neurovascular Diseases: Potential Implications for Transfusion Medicine. Transfus Med Rev. 2024;38(4):150858. Epub 20240918. doi: 10.1016/j.tmrv.2024.150858. PubMed PMID: 39413667.
6. Raza S, Callum J, Modi D, Sztainert T, Shih AW, Schull MJ, et al. Canadian donations and transfusion database (CANDAT): From blood donors to transfusion recipients. Transfusion. 2025. Epub 20250421. doi: 10.1111/trf.18257. PubMed PMID: 40255123.

1,776
The Issue
Dear Decision-Makers,

I ask that the Public Health Agency of Canada (PHAC) rescind its decision to sunset the Blood Safety Contribution Program (BSCP) – namely its Transfusion Transmitted Injuries Surveillance System (TTISS) and the Transfusion Errors Surveillance System (TESS) – effective April 1st, 2026 (as the arbitrarily-selected de-activation date).
This unilateral decision was issued on August 28th, 2024 without engagement of patients or the nation-wide transfusion service community supporting them. Only a handful of those who directly contribute data to the programs were notified. Lacking any avenues for meaningful negotiation or permissions for fan-out, this was an effective silencing of potential dissent and action.
On its website in the months that followed, PHAC did not reveal its decision, though the (anonymized) letter was ultimately exposed by the counter-action of the Canadian Society for Transfusion Medicine (CSTM) in their Hemovigilance Under Threat webpage https://transfusion.ca/our-work/hemovigilance/ ; letter: https://transfusion.ca/wp-content/uploads/2025/12/PHAC-Sunsetting-Announcement-De-identified.pdf on May 28th, 2025 prior to their annual conference.
A Call-to-Action open letter by Shih & Prokopchuk-Gauk, https://nacblood.ca/en/resource/nac-letter-cbs-ptblc-urgent-need-transform-canadian-hemovigilance-system-call-action from the National Advisory Committee (NAC) articulated numerous concerns on this decision.
On July 25, 2025, the leaders of Canada’s blood systems—Dr. Graham Sher of Canadian Blood Services and Nathalie Fagnan of Héma‑Québec—issued a joint letter urging reconsideration of the decision to close the BSCP. They proposed measures to address the findings that led to the shutdown plan, and called for a moratorium on decommissioning steps until formal discussions and actions on void management are successfully completed. https://www.transfusion.ca/getmedia/bc7e5d70-55c1-4163-8875-afa69cfa2349/2025-07-25_Letter_CEOs-to-PHAC-President-Nancy-Hamzawi_re_Consensus-Conference_EN.pdf.aspx
If the data infrastructure, its holdings, and the staffing of TTISS are altogether made obsolete, we are left to rely on the residual Blood Regulations in the Canada Vigilance Program (CVP) for counting a more limited range of adverse events (AE) or errors/accidents (E/A) of interest. The focus drops to container-side (supplier-relevant) concerns rather than to the full end-to-end process of transfusion safety and/or to the capture of extraordinary breakthrough injuries unique to a product-patient interaction (however unimpeachable the donor/blood and transfusion safety practices might have been).

Put simply, we are violating our Public Health responsibility to sense known or emerging harms by this top-down devaluation.
This betrays public trust, while backsliding on the global stage of meeting basic transfusion safety standards.
Hemovigilance aims to capture the observed harms of transfusion to make this common form of care safer. We code for what the harm was, its severity, the persistence of an injury’s effects (for those who survive), and for whether the event has implications for harm to others (ie- were there missed clues in the past; are there current containers to quarantine or recall now; and are there changes to future donor management or production practices).

The blood operators/manufacturers and the CVP have coordinated well on many “3D hits” in blood quality, be it a transfusion transmitted infection (TTI) or another reliably toxic factor.
However – not one of these core bodies is looking comprehensively at all the other now dominant and enduring harms that TTISS has been tracking. While not usually related to specific donors or modes of production/preparation, these are some of the greatest threats appreciated in transfusion care today (be it by frequency, severity, and/or by the power to prey upon the most fragile of our patients).
#1: Transfusion-associated circulatory overload (TACO). [1] Globally, this is the #1 cause of transfusion-related death. When TACO is re-scaled for under-reporting, it is also among the commonest of transfusion reactions (at a 1-10% rate per blood encounter). TACO is notoriously “disposition-escalating” (ie- summoning critical care teams or transfers [in 20% of cases][2]), and protracted in its effects (ie- extending hospital lengths of stay).
TACO is of such importance in the eyes of laboratory assessors that it has been the subject of prime inspection (per Bulletins on the expectation that policies identify those at risk and provide mitigation options).[3] TACO reflects problems upstream of the integrity of what sits within the container, and there are greater clinician, systems, and patient pain-points driving this often preventable event. TACO targets those with already poor heart-lung function, with the most susceptible at the age-extremes (the smallest, sickest, and/or oldest of our patients).[4] Closing TTISS and TESS is a curtain call for knowing the counts, understanding contributors (eg- under-resourced wards [insufficiently-examined patients, poorly paced infusions]), and designing course-corrections.
#2: HyperHemolysis Syndrome (HHS) as a particular kind of Delayed Hemolytic Transfusion Reaction (DHTR).
Tracking with the devastation of sickle cell disease (SCD) are the compounding weights of systemic racism and complex blood biology (ie- red and white cell co-adaptive variants selected by tropical infectious diseases like malaria). The latter variants can make transfusions trickier, no matter how meticulous our matching approach may be.
HyperHemolysis Syndrome (HHS), an unexpected immune over-reaction to transfusion, leaves a blood recipient more anemic than where they started. Red cell destruction goes beyond the offending unit with demolition of the patient’s own red cells, while the power to compensate is also undermined. A multi-system injury, despite intensive care, may nevertheless end in death. At hospitals taking care of people with SCD, fatalities from HHS can be on par with deaths from TACO or non-overload transfusion-related acute lung injury (TRALI).
If TTISS closes, there is no one left to keep central count of HHS.
This contravenes all united calls by exemplar hemovigilance systems, such as the Serious Hazards of Transfusion in the United Kingdom [SHOT-UK] www.shotuk.org/wp-content/uploads/2024/10/SHOT-Bite-No.15-Hyperhaemolysis.pdf and advocates within the AABB. https://www.aabb.org/news-resources/news/article/2023/10/16/aabb23--hyperhemolysis-syndrome-presents-challenges
Without learning as much as we can from each case and their collective features, we cannot make organized progress in this already-marginalized population.
To suppress hemovigilance of HHS is to amplify rather than redress a major existing inequity.
The reporting of red cell rejection events across a spectrum – from minor delayed serologic transfusion reactions (DSTRs) (asymptomatic antibody responses), to delayed hemolytic transfusion reactions (DHTRs) (partial to complete anti-unit effects), to HHS (life-threatening bystander cellular effects) – has illuminated the impacts of insufficient laboratory information centralization as well. TTISS has been a repository for this continuum of reactions, and has therefore been a systems-integration "indicator."
The failure of laboratory information handover allows the unintended gamble of provocative re-exposure to an offending antigen in a next transfusion. The strategic neglect of healthcare fragmentation-related transfusion injuries is a means by which to suppress evidence for investing in better health information technology and its inter-operability.
#3: Other events not related to product quality, but rather to the biology of the host, and/or host-donor (idiosyncratic) interactions:
Severe, high case-fatality-rate adverse events such as post-transfusion purpura (PTP) and transfusion-associated graft versus host disease (TA-GVHD), when they are not due to operator or dispensation service failures, also go silent if TTISS closes.
How do we count the emerging concern of possible cerebral amyloid angiopathy?[5]
A national loss in tabulation-power for any such reported events-of-concern is incomprehensible to jurisdictions with developed hemovigilance systems.
The operating costs of the BSCP have not only been trivial in the context of overall government spending (ie- at just over $2million CAD annually – a mere nickel for each Canadian), but diminishing over time. Participating hospitals have long financed the largest part of their own reporting workload.
Relative to the costs of the entire blood system (at >$2billion CAD annually), TTISS costs < 0.1% – a figure that is also half of what it once was.
TTISS, despite its progressive underfunding and now the existential threat against it, gets credit for being the most informative data house (relative to the federal offices that would remain).
TTISS, operated (as with the health care systems) on the provincial and territorial level, has its very origins rooted in the evidence and recommendations of Horace Justice Krever in the Royal Commission of Inquiry on the Blood System in Canada (“the Krever Commission”).
As the nation sits upon literal mountains of data, and just as visions of better-connected, vein-to-vein Big Data are dawning with the promise of new analytics and enhanced insights in personalization,[6] the system is nearing elimination rather than enhancement.
Many Canadians still recall the tainted global blood tragedy.
Dr Graham Sher, CEO of Canadian Blood Services, wrote this at the passing of Krever 2 years ago in 2023:

Granted the triumphs against viral TTIs, and improved platelet and plasma safety by the contemporary modifications of pathogen reduction and additive solutions, would Krever agree that we have made such strides that a secretive shut-down of the BSCP by PHAC is justified?
How or why are other less-evidence-powered, non-commission-inspired programs permitted greater inefficiencies or investments?
How do we square the moves of an allegedly pro-healthcare administration with the playbook tactics of more conservative/opposition-party platforms that the majority of Canadians voted against?
I agree with the NAC Letter in the call to conserve (or enhance) hemovigilance funding, with a commitment to improve our system by reorganization, rather than by scripting its collapse.
This would align better with (rather than thwart) the recommendations on sharpening roles and responsibilities, with more timely analytic turnarounds, per the evaluation report of 2023. https://www.canada.ca/en/public-health/corporate/transparency/corporate-management-reporting/evaluation/blood-safety-contribution-program-2017-2018-2021-2022.html
Dismantling event or error/incident reporting systems is deeply counterproductive at a time when jurisdictions are more attuned than ever to these risks and the imperative to mitigate them.
As a fellow patriotic Canadian, each of us with a lifetime odds of 1-in-2 for needing a blood transfusion, I trust that we want what is safest for each other and our loved ones.
Let us save hemovigilance in Canada.
References:
1. Bulle EB, Klanderman RB, Pendergrast J, Cserti-Gazdewich C, Callum J, Vlaar APJ. The recipe for TACO: A narrative review on the pathophysiology and potential mitigation strategies of transfusion-associated circulatory overload. Blood Rev. 2022;52:100891. Epub 20211002. doi: 10.1016/j.blre.2021.100891. PubMed PMID: 34627651.
2. Lieberman L, Maskens C, Cserti-Gazdewich C, Hansen M, Lin Y, Pendergrast J, et al. A retrospective review of patient factors, transfusion practices, and outcomes in patients with transfusion-associated circulatory overload. Transfus Med Rev. 2013;27(4):206-12. Epub 20130926. doi: 10.1016/j.tmrv.2013.07.002. PubMed PMID: 24075097.
3. American Association of Blood Banks - Association Bulletin #15-02: Transfusion Associated Circulatory Overload (TACO). 2015 [updated 12 February 2016]. 15]. Available from: http://www.aabb.org/programs/publications/bulletins/Docs/ab15-02.pdf#search=bulletin%20%2315%2D02
4. McVey MJ, Cohen R, Arsenault V, Escorcia A, Tasmin F, Pendergrast J, et al. Frequency and timing of all-cause deaths in visits involving suspected transfusion reactions, and the significance of cardiopulmonary disturbances. Vox Sang. 2021;116(8):898-909. Epub 20210226. doi: 10.1111/vox.13086. PubMed PMID: 33634884.
5. Muir RT, Callum JL, Yu AYX, Kapral MK, Swartz RH, Black SE, et al. Beta-Amyloid Related Neurodegenerative and Neurovascular Diseases: Potential Implications for Transfusion Medicine. Transfus Med Rev. 2024;38(4):150858. Epub 20240918. doi: 10.1016/j.tmrv.2024.150858. PubMed PMID: 39413667.
6. Raza S, Callum J, Modi D, Sztainert T, Shih AW, Schull MJ, et al. Canadian donations and transfusion database (CANDAT): From blood donors to transfusion recipients. Transfusion. 2025. Epub 20250421. doi: 10.1111/trf.18257. PubMed PMID: 40255123.

1,776
The Decision Makers
Supporter Voices
Petition Updates
Share this petition
Petition created on June 25, 2025